Cell counting is a routine method that is performed to determine the level of cell concentration in a sample. In this way, the growth behaviour of a cell line can be observed if a certain cell count must be reached to perform an experiment or to check the influence of different factors on growth behavior.1
Since this counts a frequently performed process in everyday laboratory practice, it is important for the procedure to be as simple and time-saving as possible while providing reliable results, as it forms the basis of many experiments.
The fluidlab R-300 offers a fast method of counting cells automatically. Due to the comfortable display size it also ensures high statistical certainty.
The most important features of the fluidlab R-300 in one glance
The fluidlab R-300 is the first truly portable automated cell counter. While other automated cell counters are often heavy, large in size and therefore bound to a fixed place in the laboratory, the fluidlab R-300 is not only smaller but also significantly lighter.
This enables the implementation of cell counts at different work locations in a space-saving way – for example directly beneath the sterile laboratory bench.
Other advantages are:
- the large "field-of-view" (5.3 mm²) and automated determination of the cell count reduce human error and increase statistical certainty
- the holographic microscope does not need to be calibrated
- due to automatic focus the focusing level does not have to be selected by the user
- a viability measurement can be performed without the use of dye
This makes the counting of cells in the laboratory easier and faster at the same time.
Manual vs. automated cell counting
In many research institutions and universities, cell cultures are counted manually. Manual cell counting is performed using counting chambers also called haemocytometers. After the cover glass has been placed on the counting chamber and the sample liquid has been applied, the cells are then counted under the microscope.2
Based on the counted cells, it can be calculated how many cells per volume are contained in the sample. From this, a simple calculation can be made to determine the cell concentration in the initial solution.
If the counting is performed manually and a distinction is to be made between living and dead cells, the trypan blue staining is typically used in addition. The dye stains dead cells with permeable cell membrane blue, while living cells remain unstained. In this way, the proportion of living and dead cells, the so-called viability, can be determined. The manual methods for the determination of cell count and viability are very elaborate and time-consuming thus often subject to errors.
Alternatively, there is a wide range of automated cell counters which do not necessarily use the same method. An automated method is the determination of cell counts via microscopy. Here, a software counts and characterizes cells in a microscope image.
The fluidlab R-300 also utilizes this procedure. A new microscopy technology is used here that makes use of holograms. The automatic counting is much faster giving the advantage that several samples can be counted one after the other, leaving additional time for further work. Furthermore, automated counting devices can determine additional measurement parameters, such as the size distribution of cells in the given suspension.
What influences the result of a cell count?
There are various factors that can negatively influence the result. These can be of different nature and can occur even before the actual measurement is carried out. The fluidlab R-300 cannot eliminate them completely but the device can minimize them.
Some of them can have a negative effect on counting with both an automatic cell counter and a counting chamber. These include the following:
- Poor mixing of the sample before measuring leads to falsified results. If the suspension has not been properly resuspended before the measurement is performed, the counting may not give an authentic result since it does not reflect the correct distribution of cells within the suspension.
- The presence of air bubbles, which can occur when filling the counting chamber also complicate the cell counting. Manual counting must be interrupted and the bubbles must be removed first if they impede counting.
Since manual cell counting offers considerably more potential sources of error, the following factors can additionally influence the result negatively when this method is being used:
- Lack of attention due to fatigue or eyestrain as well as distraction can lead to deviating results.
- Subjectivity when counting manually can lead to different results: The distinction between cells and other particles under the microscope is not always easy and can vary from person to person. This can lead to different results while using the same sample. Cell fragments (debris) can especially lead to uncertainties.
- The incubation time of the trypan blue for manual cell counting must be controlled carefully as the dye is cytotoxic. If the counting takes too long, more and more cells will die. Consequently, more and more dead cells are counted and the result is falsified.
Handling errors such as poor mixing of the suspension cannot be eliminated by the R-300. However, the instrument carries out the counting automatically, thus eliminates faulty results due to lack of concentration.
Furthermore, the neural network of R-300 can not only distinguish cells from other particles, but also distinguishes dead cells from living cells. This makes staining with trypan blue unnecessary.
The acella sample carrier does not have to be assembled and is ready to use immediately, which speeds up the counting process overall. The standardized chamber height of acella makes individual counts of a sample more comparable and reproducible.
High-resolution microscope with new technology: The advantages of cell counting with the fluidlab R-300
The fluidlab R-300 is calibrated at the factory and requires no further calibration by the user. Unlike many other automated cell counters, the R-300 is small and portable. This allows cell counting directly under the bench. In addition, it takes up only a small portion of space on the working surface.
The neural network counts cells reliably with a high statistical certainty. It distinguishes cells from other particles such as cell debris or cell clumps.
Both cell counts and viability measurements normally require time-consuming sample preparation. With the R-300, many samples can be measured in rapid succession. By omitting the cytotoxic dye, the sample also remains stable for a longer period of time.
The individual cells are extracted from the background. The neural network then analyses the cells cut out from the background by distinguishing between cell forms and measuring the cell's viability.
Viability is determined by the neural network and is based on a signal proportional to the intracellular refractive index. In this case, this parameter is linked to the protein concentration of the cell. In addition, measurement parameters on the morphology of the cell are included in the evaluation.
In this way, the measurement of viability is not only faster and easier to perform, but also cheaper and more specific than the standard method. Thus quality management in the cell culture laboratory can be simplified.
To perform cell counting with the R-300, the cells must first be in suspension in a suitable dilution. The cell count application of the R-300 is particularly suitable for counting the cells after they have been washed. After preparing the sample, the sample carrier adapter 01 is inserted into the fluidlab R-300 and the sample carrier acella is filled.
The Cell Count and Viability applications can be selected by tapping on the main screen. In the input mask the measurement can be named and the cell size and dilution can be defined.
To learn more about our fluidlab R-300, please contact our sales team at anvajo.contact@gmail.com.
What kind of equipment is needed for the analysis?
To perform a cell count, the fluidlab R-300 and the sample carrier acella with suitable adapter 01 need to be used. The device is already equipped with the appropriate software. It is not necessary to purchase additional licenses. You can start your measurement right away.
How does the fluidlab R-300 output the measurement result?
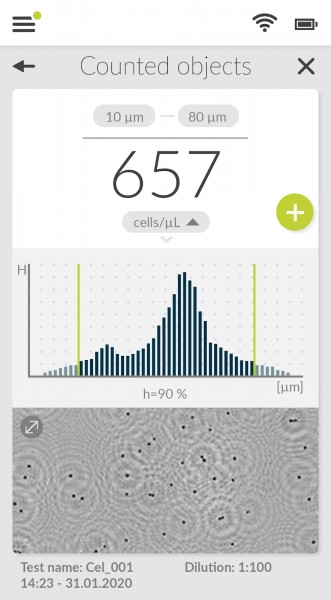
After counting, the fluidlab R-300 displays the results as "counted objects". The concentration is shown in cells/µL. However, we are planning to give the option for displaying the concentration in cells/mL in the future.
Below the given result you will find a histogram in which the cell size is plotted against the cell number. This makes it easy to determine how many cells of a certain size were found.
The green borders left and right in the histogram can be moved arbitrarily so that the cell count is only displayed within the selected size range. The recognized cells are then marked with green boxes.
In which departments can a cell count be performed with the fluidlab R-300?
The fluidlab R-300 is CE-certified and must therefore be individually matched to the requirements with regard to further quality criteria that must be fulfilled for use in some specialist areas. The fluidlab R-300 is not approved for use in clinical diagnostics and may only be used for research purposes.
In university institutes for biology or medicine the R-300 can be used in
- laboratory internships
- departments of cell and molecular biology
- departments for biotechnology
- departments of plant physiology
- departments for ecology
- departments for microbiology
- departments of biochemistry
- department of toxicology
There it can be used for cell counting of permanent and primary cell lines, algae and yeast cells.
In university institutes of medicine it is used for basic and applied research. There it can also be used for cell counting and viability determination.
Furthermore, the R-300 can be used in biotecnological companies to count various cell cultures.
Small, accurate, cost-efficient: What does the cell counter fluidlab R-300 by anvajo cost?
The fluidlab R-300 combines two devices in one. This results in a significant price advantage, since it is not only a cell counter but also a spectrometer. Cell count and the measurement of viability can be performed without big effort. Pictures of the sample can be taken due to the holographic microscope. Furthermore, the neural network recognizes and quantifies cells with a high statistical certainty. Countings can be performed at different workplaces since the analyzer is a small and portable device.
Scope of delivery of the fluidlab R-300 cellcounter
To start immediately with the measurements the fluidlab R-300 comes in the anvajo fluidlab R-300 Set, containing
- the fluidlab R-300
- one key for the adapter
- the adapter 01 for the sample carrier, acella
- the adapter 02 for scattering measurement
- the adapter 03 for absorption measurement
- one sensor cleaner
- one power supply unit
- one USB charging cable
- one user manual
- one Quick-start Guide
No further equipment is required and you can begin immediately with your measurements.
Innovation for your workplace: Increase the quality of your cell counts
Automatic cell counting reduces potential sources of error during counting to a minimum.
Thanks to the intuitive handling of the device and the automatic counting by the neural network, the determination of the cell concentration is made much easier.
Moreover, steps such as staining the cells with trypan blue to determine viability are completely unnecessary.
If you decide to purchase the fluidlab R-300, discounts or benefits can be granted, if the order is for universities, institutes or companies and a higher quantity has been ordered.
However, this is done according to an individual agreement, as there are no flat-rate discount groups.
Are you interested in purchasing several devices for your research group, internships or company locations? Contact us for a personal offer atanvajo.contact@gmail.com.
Since the software is always automatically pre-installed on the device, it does not need to be purchased or extended. Similarly, no additional licenses are needed.
Find out more about the fluidlab R-300 on our homepage or contact our sales team.
________________________________
1 Schmitz, 2011. Der Experimentator: Zellkultur. 3 Hrsg. Heidelberg: Spektrum Akademischer Verlag.
2 Vgl. Schmitz, 2011